Consolidated CDA merged onto the scene with the announcement of the final rule for Meaningful Use Stage 2 in February 2012. A term unheard of by most before the final rule, C-CDA was an attempt to correct some of the difficulties of the CCD version mandated for Meaningful Use Stage 1. As of 2014, Consolidated CDA remains a Draft Standard for Trial Use, which has drawn some criticism, but it still offers many advantages over standards used in Meaningful Use Stage 1.
Consolidated CDA is a compilation of nine different types of documents, including a revised CCD, and is utilized to construct the summary documents for Meaningful Use Stage 2. This paper serves to untangle some of the terminology around Consolidated CDA, including answering:
- How is Consolidated CDA different from CDA?
- How is CCR related to CCD and CDA?
- Summary of Care, Patient Summary, Clinical Summary and Export Summary: What are they?
By taking a look back at how the health IT industry got to where it is today, it becomes easier to understand the terminology and how all the different document types are related.
Meaningful Use Stage 1
From the perspective of sharing documents, Meaningful Use Stage 1 was focused on making sure that vendors had the technology in place to send and receive electronic documents. Eligible hospitals and eligible physicians were required to prove that they had the capability to send and receive documents by doing it once, but were not required to send any documents on an ongoing basis. With most facilities scrambling to meet just the minimum requirements for all Meaningful Use criteria, few facilities went beyond the minimum requirement of just proving that they could send documents.
When sending documents, Meaningful Use Stage 1 allowed for two different document types that could be utilized:
- CCR Continuity of Care Record
- CCD Continuity of Care Document
CCR is an ASTM standard, while CCD is an HL7 standard and part of the CDA (Clinical Document Architecture) document family. These three acronyms (CCR, CCD, and CDA) are sometimes referred to as the three Cs of healthcare. It is useful at this point to take a step back and review the health IT history of events that led us to these document types.
Three Cs of healthcare
One might wonder how the health IT industry ended up with two valid formats instead of one for Meaningful Use Stage 1. And then how those two formats were reduced to one for Meaningful Use Stage 2. Having knowledge of the historical decisions that were made might shed some light on how the industry ended up where it is today.
CCR
Since before the use of electronic documents, facilities have been trying to determine the appropriate information to be included in a patient’s medical history as they are transferred from one facility to another. One of the most notable attempts to do this was by the Massachusetts Department of Public Health, which had a three-page paper-based Patient Care Referral Form that was in widespread use for many years in Massachusetts.
Based on the success of this paper-based form, several key agencies combined resources to create an electronic version. The agencies jointly developing the electronic format included ASTM International, the Massachusetts Medical Society, HIMSS, and the American Academy of Family Physicians. It is intended to do in electronic format what had been done so well with the three-page paper-based form. Its intent was to assure a minimum standard of health information transportability when a patient is being transferred to another provider.
This activity produced the CCR standard, which was published in January 2006. The new standard was backed by the CCR Acceleration Group, which was an industry alliance of approximately 75 companies. In general, CCR had more of its support from the mid-tier and smaller vendors and institutions.
CDA
At the same time CCR was being developed, the HL7 organization was busy working on developing their family of electronic documents called CDA. CDA Release 2 unveiled its normative edition in May 2005. One of the implementation guides for CDA Release 2 was the Care Record Summary that included specifications for summary of care information.
Unlike CCR, CDA was a framework that could be applied to a variety of workflows, not just summary of care. From History and Physical documents to Radiology Reports, CDA would provide a selection of different document types that could be applied based on the appropriate needs for the given scenario.
HL7 had much recognition in healthcare IT for developing standards. In particular, HL7 v2 is firmly entrenched in the healthcare setting in the United States for applications to share data. CDA was part of the new HL7 v3 initiative, which at the time was touted as the next generation replacement for HL7 v2 standard.
CCD
Eventually HL7 and ASTM got together and combined resources to deliver to the industry a new summary of care document, they called it CCD. It was advertised as being the best of both worlds. It had the same content as a CCR, which was proven to be effective as a paper-based solution. However that content was fit into the framework of the CDA, so that it would share similar structure with all other document types in the CDA family. Once an application has been designed to produce and consume a CCD document, it should be a much smaller step to incorporate other types of CDA documents structured in a similar way. The first release of CCD was made available in April 2007.
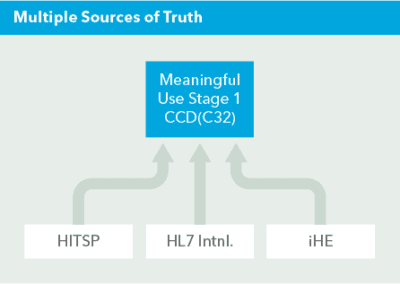
Multiple sources of truth
Even though CCD was advertised as the summary of care document for health IT moving forward, when Meaningful Use Stage 1 was released—to the surprise of many—CCR was still included as one of the possible document standards. As mentioned already, since the exchange of these documents was not required to be achieved more than once, Meaningful Use Stage 1 did not do much to propagate the use of either CCR or CCD. However, because applications were required to have the capability to support the transmission of both CCR and CCD, much was learned from application vendors about the ease that these documents can be supported. The feedback from the industry was that the creation of a conformant CCD and the ability to consume that document as structured data proved very challenging.
The CCD document included in the Meaningful Use specification for Stage 1 was more specifically CCD as described in the Health IT Standards Panel (HITSP) interoperability specification C32. C32 is a constraint, essentially a set of additional rules to follow, in addition to the original implementation guide published by HL7. And in addition, the C32 constraint referenced additional specifications. There were several guides/sets that had to be referenced to fully understand the C32 specification. They included:
- HL7 CCD
- IHE’s PCC XPHR profile
- HITSP’s C32
- C83 Data templates
- C80 Vocabularies/coding systems
This created an overly complicated standard with convoluted pointers to other documents.
Subsequently, each individual rule lookup might potentially have several reference points found across various technical volumes. This became unmanageable for applications to build robust interoperable document interfaces that could truly share data in a consistent and reliable way. The term ‘multiple sources of truth’ was used to describe the dilemma of trying to navigate the standard.
Meaningful Use Stage 2
The document requirements for Meaningful Use Stage 2 served to eliminate some of the challenges noted above with Meaningful Use Stage 1. The major shift was to consolidate all of the various documentation for CCD, as well as other document types, into one implementation guide, the C-CDA implementation guide.
CDA was originally created as a framework, with each individual document type having its own implementation guide(s). Consolidated CDA is not a new standard, but rather a streamlining of the existing CDA standard—compiling all of the most common CDA document types into one implementation guide.
This one new implementation guide would serve as a reference for all the document summaries utilized in Meaningful Use Stage 2, with the goal of drastically decreasing the complexity for applications to meet the requirements to produce and consume these documents.
What is Consolidated CDA?
The C-CDA implementation guide contains the specifications for nine document types within the CDA family. They include:
- CCD
- Consultation Note
- Diagnostic Imaging Report
- Discharge Summary
- History & Physical
- Operative Note
- Procedure Note
- Progress Note
- Unstructured Document
Consolidated implementation guide
One of the main objectives behind C-CDA was to eliminate the multiple sources of truth. This was accomplished by moving to one implementation guide, rather than having to sort through multiple specification documents from HL7, HITSP, and IHE. The result was the Implementation Guide for CDA Release 2.0 Consolidated CDA Templates, or in short referred to as the Consolidated CDA Implementation Guide.
Another requirement that emerged around the same time was the need for this implementation guide, which is mandated by Meaningful Use, to be made freely available. The result was not only one implementation guide, but it was also available to everyone who wanted to use it at no cost.
Bucket of reusable templates
Another concept that was employed with C-CDA was the idea of having a bucket of reusable templates. The idea was that any document within the C-CDA family would use the same template for a given section or entry as any other document. This eliminates “recreating the wheel” and allows applications to easily move to supporting more than just one document, because once the work is done for one document type it should be highly leverageable for other document types.
For example, a vital signs section can be included in both a CCD and a progress note. If an application has built out the structure for a vital signs section in CCD, then they should immediately be able to add the same vital signs section to a progress note. There should be no difference in the way vital signs are coded among the CDA document types because all CDA documents will utilize the same vital signs template.
In Figure 1, the box with the blue header represents the bucket of reusable templates for the sections in CDA. A CCD document has 15 available sections that can be included. Of those 15, five can also be included in a progress note, including the vital signs section. For the common section types, templates ensure that they will be encoded exactly the same among the two documents.

Templates exist at three levels (see Figure 2):
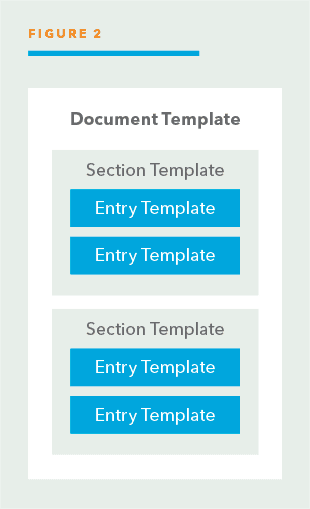
- Document Each of the nine document types included in the C-CDA standard has its own template to define the sections that are included in the document.
- Section As seen in Figure 2, each section is templated to maintain consistency of how the same sections are coded across the different document types.
- Entry Specific entries within each section, such as medication dispense, are templated as well.
The C-CDA Implementation Guide provides all the specifications needed to comply with the documents required in Meaningful Use Stage 2. The objectives that include these documents are discussed further in the following section.
What objectives utilize Consolidated CDA?
Meaningful Use requires eligible hospitals (EHs) and eligible professionals (EPs) to meet objectives to pass Meaningful Use. There are core objectives, which are required, and menu objectives, for which the EH and EP can chose from a list. Only a limited number of objectives are related to the use of C-CDA documents.
There are two core objectives for eligible hospitals (EHs) that utilize a C-CDA document and three core objectives for eligible professionals (EPs). They are as follows:
Transfer of care (summary of care)
- EP objective: When transitioning or referring their patient to another care setting, the EP should provide a summary of care record for each transition of care or referral
- Measure: more than 50% of transitions of care and referrals with 10% sent electronically
- EH objective: Same as EP
View, download, and transmit (patient summary)
- EP objective: Provide patients the ability to view online, download and transmit their health information within four business days of the information being available to the EP
- Measure: access to more than 50% with more than 5% actually accessing
- EH objective: Provide patients the ability to view online, download and transmit their health information within 36 hours after discharge from the hospital
Clinical summary
- EP objective: Provide clinical visit summaries for patients for each office visit within one business day
- Measure: provide for more than 50%
For the Transfer of Care core objective, the Summary of Care record described in the objective is a C-CDA document. This document should provide all the relevant information necessary to continue care for the patient as they progress from one facility to another.
For the View, Download, and Transmit core objective, the Patient Summary document available for downloading and transmitting by the patient is a Consolidated CDA document. This objective gives the patient control of their medical information. The patient summary document allows the patient or their caregiver to review and confirm the medical information from their provider, and also gives them control to forward this to other providers they have visited or plan to visit.
For the Clinical Summary core objective, the Clinical Summary document utilized for this objective is a C-CDA document. The idea behind this objective is to share with both patients and family members what occurred during office visits to create awareness and be used to assist in care coordination.
Data portability (export summary)
In addition to these objectives, EHR applications must have the ability to enable a user to electronically create a set of summaries that contain the most current clinical information for all patients in the EHR application. This Export Summary must be formatted according to the C-CDA Implementation Guide. This is the Data Portability requirement for EHR applications to support, and allows for a data dump, if required, in a structured format.
How is C-CDA structured for Meaningful Use?
In Meaningful Use Stage 1, specific document types were utilized: CCR and CCD. These document types contained specific sections of clinical data. For Meaningful Use Stage 2, none of the nine specific document types called out in the C-CDA Implementation Guide are utilized exactly as they exist.
As mentioned in the previous section, there are four different types of document summaries utilized in Meaningful Use Stage 2:
- Summary of Care
- Patient Summary
- Clinical Summary
- Export Summary
Each of these document summaries is based on the C-CDA standard. But, each has its own unique requirements as to what should be included in the summary based on the purpose of the summary, such as transitioning care.
In general, these summary document types include the following categories of clinical data:
- Care Planning
- Care Team Information
- Cognitive and Functional Status (not included in Patient or Clinical Summaries)
- Conditions or Concerns
- Encounter Information
- Medications
- Immunizations (not included in Patient Summaries)
- Observations and Results
- Patient Information
- Procedures
However, the details as to what is included within each one of these categories varies for each of the summary types. For example, date of visit is included as Encounter Information for a Clinical Summary since it refers to a specific visit in an ambulatory setting. On the other hand, date of visit is not included in the other summaries since they are based on a snapshot in time.
For full details on what specific data is included in each of the specific summary documents, please refer to the Companion Guide to HL7 Consolidated CDA for Meaningful Use Stage 2 produced by the ONC. It is a comprehensive document that outlines all the details for creating valid C-CDA documents for Meaningful Use Stage 2.
Conclusion
The goal of implementing C-CDA in Meaningful Use Stage 2 was to simplify the learning curve to understand the structure of the required documents. Consolidating all the documentation under one implementation guide along with the use of templates has gone a long way in achieving this goal. Extra complexity was added by introducing four different types of summary documents for the different requirements of Meaningful Use, but this was deemed necessary to provide only the required clinical information necessary for each use case.
However, C-CDA has not been without its critics. In October 2014, the American Medical Association released a document that expressed concerns over C-CDA and its ability to foster interoperability. The document stated that the ONC mandated its use in Stage 2 even though it had “very little real world testing, nor was it balloted or approved for standardization by HL7” and because of this had “wild variation in technology versioning.”
All in all, it has been a large improvement over the struggles of Meaningful Use Stage 1, but still at this time is in need of a final consensus so that developers can be working from one consistent, finalized standard.
